
-
Over three decades of proven pharmacology experience
We have a proven track record of developing and manufacturing SDDs to overcome poor drug solubility and delivery.
-
Overcoming challenges
With our know-how, technologies & facilities, we address critical formulation issues quickly from pre-clinical to clinical evaluation.
-
Rapid volunteer recruitment and outstanding retention
Over the past 2 years, we have advanced more than 25 spray-dried dispersion (SDD) formulations into clinical studies.
-
Need to delete and check
We recognize the need to move rapidly and efficiently through clinical development. So we ensure a seamless transition to larger-scale manufacturing and drug product commercialization by leveraging our expertise to scale up manufacturing processes from Phase I to meet the demands of later clinical trials..
Benefits of spray dried drug product formulation strategies
Quotient Sciences capabilities for pharmaceutical spray drying help reduce development risks and simplify the drug development supply chain. We can help you:
- Identify and address drug product formulation challenges quickly, and support your drug product formulation development from preclinical through clinical evaluation
- Select best-fit SDD components and process for the intended final dosage form—this includes the solvent selection, API loading, polymer selection, and spray parameters
- Manufacture GMP supplies of lead SDDs and dosage forms
- Optimize the performance of your spray dried formulation through clinical testing—leveraging our Translational Pharmaceutics® approach for integrated drug development
- Provide ongoing drug product supply for your Phase II proof-of-concept studies and beyond

Our Spray drying formulation development expertise

More than 500 programs of 31,500+ volunteers
completed in therapy areas including oncology, neurology (CNS), and pediatrics.

Trusted for over 17 years
ranging small batch sizes to support clinical trial materials, to over 500 kg for solid oral dosage forms in commercial drug product.

100+ customers
globally have used the Translational Pharmaceutics® platform.
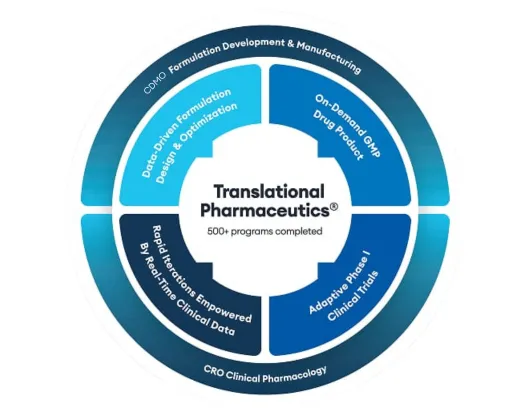
Quotient Sciences Translational Pharmaceutics® platform
Our drug product and clinical pharmacology capabilities combine into a flexible drug development and manufacturing program: Translational Pharmaceutics®.
Quotient Sciences Translational Pharmaceutics® platform helps you accelerate to and through first-in-human trials and optimize formulations coming out of Phase 1 studies, with the ability to release and dose in our clinics in under seven days, adapt to emerging clinical data in real time, and more.
Contact us to learn more about the benefits of integrated Phase 1 clinical studies and drug product services with Translational Pharmaceutics®.
Quotient Sciences is a global accelerator of drug development, with services spanning contract research, development, and manufacturing that make us more than just a traditional CRO or a CDMO.
Our CRDMO offering includes:
More insight, direct from our customers:
"Quotient Sciences has worked with us on several pediatric projects and has been very effective in translating our product concepts into successful prototype formulations."
-Testimonial from a UK-based pharmaceutical company specializing medicines for children
"Quotient Sciences has reduced our time-to-clinic and time-to-market. They understand the issues in drug development and have taken us from early stage to commercial in the most efficient way."
-Testimonial from a former specialty pharmaceutical company
"Quotient Sciences’ expertise in improving the taste, smell, and texture of oral formulations makes them the ideal partner for our pediatric clinical programs."
-Testimonial from a global pharmaceutical company developing therapies for people with endocrine diseases
Small biotech
"Our small biotech company worked with Quotient Sciences on developing a complex formulation and validating the different iterations of the formulation in PK clinical studies using the Translational Pharmaceutics® approach. The project was successful, and we were generally impressed by the skillsets, professionalism, and courtesy of Quotient Sciences' scientists and other subject experts. We have also chosen to work with Quotient Sciences on two subsequent projects."
Biotech with infectious disease focus, acquired by a Fortune 50 pharma company
"Due to the low density and poor flow characteristics of our drug substance, we experienced significant blend and content uniformity issues. By transferring our program to Quotient Sciences, the development team was able to quickly produce demo batches through multiple blending steps and roller compaction using the Gerteis Mini-Pactor. The increase in drug load from 25% to 40% was a significant improvement as we prepared for future Phase II/III trials and this ultimately benefits the patients by reducing the pill burden. We have successfully manufactured several GMP batches of tablets at various doses using a common blend. Quotient Sciences is an outstanding drug development partner with a no-nonsense approach, excellent communication, speed, and robust, easy-to-review documentation, from analytical methods to master batch records."
Former small biotech focused on oncology
"Quotient Sciences is an honest and straight-forward company with a very progressive attitude... What makes them unique is the way in which the pharmaceutical products are made on site and transferred, without leaving the building, to the clinical unit and a good, large and reliable pool of healthy volunteers."
Clinical-stage pharmaceutical company, focused on endocrine therapies
"There is an art to the development of taste-masked oral products for pediatric indications. Quotient Sciences’ expertise in improving the taste, smell, and texture of oral formulations makes them the ideal partner for our pediatric clinical programs."
Your partner to bring new treatments to market. Fast.
From the next generation of emerging biotechs, to Fortune 100 pharmaceutical companies, we have worked with countless companies across the pharmaceutical industry over the last 30 years. Speak with one of our experts to learn more about our capabilities and how we can help you reach your program's milestones.






